|
All you need to know about the safety of lead-acid batteries. Read full article to learn:
How do lead-acid batteries work?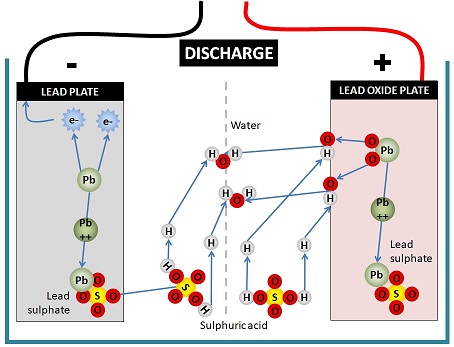
The lead-acid battery uses an electrochemical process to produce energy. In other words, it converts chemical energy into electrical energy using an electrochemical reaction between two dissimilar metals. The two parts of the battery which are in contact with the electrolyte are called “plates.” These 2 metals are: Lead Peroxide (PbO2) which is the positive pole and Sponge Lead (Pb) which is the negative pole. The electrolyte solution reacts with these 2 metals to produce energy. What is the Electrolytic Substance in a Lead-Acid Battery?
Batteries are made of an electrolytic substance, which is just battery acid. So what is battery acid and how does it work? It’s a liquid that has 30% to 50% sulfuric acid and distilled water. This helps convert chemical energy into electrical energy. Let’s dig deeper to find out more about how the components of a lead-acid battery work together to create electricity in a battery. Electricity is created when electric ions flow across the electrolyte solution from the negative plate to the positive plate, with currents going both ways – from the device being powered by the battery or charger back through the positive terminal of the battery for absorption by the negative plate. Essentially, when there’s a charge being used on an appliance or gadget, it leads over to another electric terminal in order to be recharged–and vice versa if power is being sent from charger back into an appliance or gadget. What are the dangers of batteries? Now that you know what batteries are made of, it’s easier to understand the risks involved. These risks include:
How dangerous is battery acid?Sulfuric acid, battery acid, is an inherently dangerous substance.
Find High Quality Batteries Now >> from https://tomaltaandbeyond.wordpress.com/2022/10/26/278/
0 Comments
Leave a Reply. |
About Us
Archives
April 2018
Categories |

 RSS Feed
RSS Feed
